Access to medicines should start with access to knowledge
In Open Access Week we draw attention to the importance of universal access to medicines research.
It’s Open Access Week, our annual opportunity to join together, raise awareness, and take action to drive equitable knowledge sharing, and the positive societal impacts open access to knowledge will deliver.
I've been an active and vocal advocate for open access to science, throughout my career. As a rare disease clinician, I witnessed firsthand the profound, often life-saving impact of access to global research on ultra-rare conditions. I fervently believe access should be universal, easy, and free for all readers.
There are challenges in providing universal access, notably, how the costs of delivering free-to-read content will be supported, and by whom. Many individuals and organisations are working tirelessly and creatively to address the challenges. Most are logistical issues that can be solved if we are committed to solving them. For priority areas we must solve them, urgently.
Providing universal access to medicines research is an urgent priority.
Universal access to medicines research
Medicines are a cornerstone of healthcare. In 2020, 4.5 trillion doses of medicines were dispensed at a cost of $1.4 trillion—approximately 16% of the global healthcare expenditure. Access to essential medicines is included in our right to health, enshrined in international law since 1946, and is a core target of the Sustainable Development Goals.
As well as increasing access to the medicines themselves, increasing and democratising access to medicines research is crucial. There is an urgent need to decrease medication errors, adverse effects, and non-adherence, and to increase knowledge on best practices, innovations, clinical-effectiveness, and cost-effectiveness. Universal, free availability of medicines research should be a priority objective of academia, pharmaceutical companies, and healthcare.
Universal access to medicines research will save lives
Current status of access
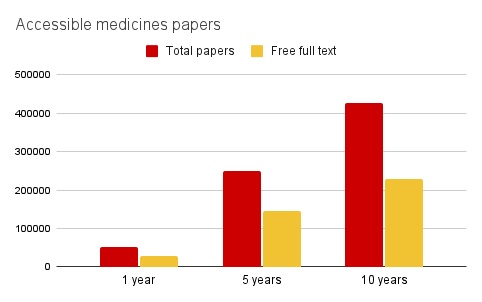
To investigate current accessibility to medicines research I used PubMed from the NIH National Library of Medicine, which is the go-to search tool for medical practitioners and researchers. PubMed gives the number of publications for a given search term, which you can filter to find those for which the ‘free full text’ is available.
I searched for papers on medicines included in PubMed over the last 10 years1 and used the full free text filter to see which were openly accessible.
Only 55% of papers had the full free text available and this has been stable over the last 10 years - very disappointing.
Lessons from Covid-19
The accepted importance of providing immediate, free access to research publications was underscored in the pandemic when more than 50 publishers made over 350,000 coronavirus-related articles publicly available in PubMed and other repositories. This was prompted by a call from scientists ‘to ensure that research findings and data relevant to this outbreak are shared rapidly and openly to inform the public health response and help save lives’ and was aligned with prior statements on data sharing in public health emergencies.
To evaluate the impact of the response, I restricted my PubMed search to Covid-19 papers2, and found that 85% were available to download, in full, at no cost.
There is a way, if we have the will
The pandemic response shows it is possible to quickly and dramatically increase accessibility of research publications. When it is considered important enough.
The Sustainable Medicines Partnership believe all medicines research should be considered important enough. We are working together to design practical solutions that will drive and deliver action.
We will share more on this in 2024.
In the meantime please checkout the excellent content on Open Access medicines research from our friends and collaborators at Oxford Pharmagenesis and Open Pharma.
Search term used in PubMed (("pharmaceuticals"[MeSH Terms] OR "pharmaceuticals"[All Fields] OR "drugs"[MeSH Terms] OR "drugs"[All Fields] OR "medications"[All Fields] OR "medicines"[All Fields] OR "therapeutics"[MeSH Terms] OR "therapeutics"[All Fields]) AND ("development"[MeSH Terms] OR "development"[All Fields] OR "clinical indications"[All Fields] OR "clinical uses"[All Fields] OR "side effects"[All Fields]))
AND ("covid-19"[All Fields] OR "coronavirus disease 2019"[All Fields] OR "sars-cov-2"[All Fields] OR "2019-ncov"[All Fields]) was added to the search above to restrict to Covid-19 publications.